Mechanical chronic low back pain results from an injury or stress on the tissues surrounding the spine, including soft tissues, muscles, bones, and joints. Often times, this type of pain is due to impaired neuromuscular control and neural inhibition of the multifidus, which is the largest stabilizing muscle in your back.
Watch the video to learn more about neuromuscular control, proprioception, and various functions of your back muscles.
Clinical Cues

Do they seem to be getting worse with time?


Do you have to shift positions or support yourself with your arms because of this pain?
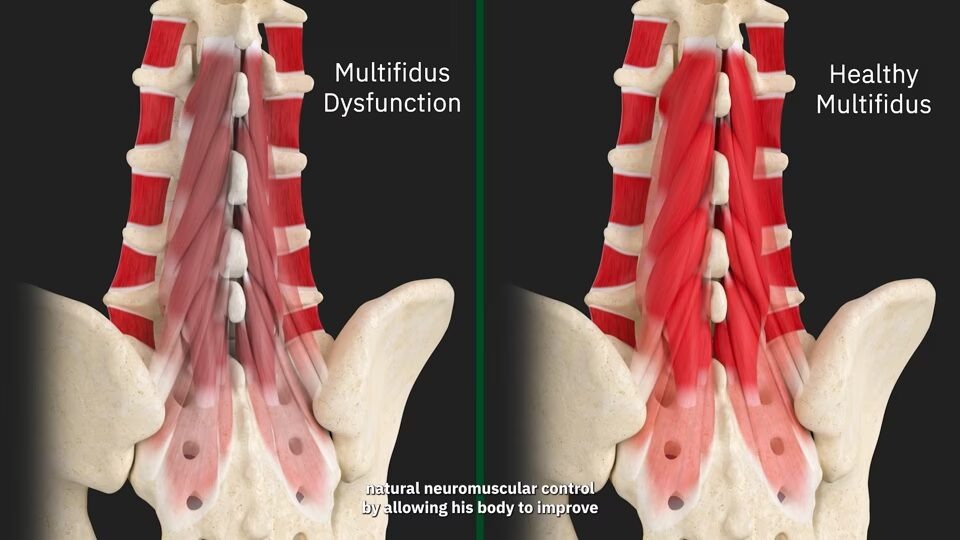
Multifidus Dysfunction can result from a loss of neuromuscular control. This can cause a lack of spinal support, leading to uncontrolled loading of spinal joints and discs, resulting in mechanical low back pain.
Watch the video to learn more about multifidus dysfunction and loss of neuromuscular control.
Restorative treatments focus on improving function for patients by addressing the underlying cause.
Palliative treatments focus on blocking pain through Spinal Cord Stimulation, oral medication, injections, or burning nerves.
Complete this survey to see if you’re a good fit
Take the first step towards finding a doctor and determining if ReActiv8 is right for you. By answering the following questions, we can provide you with general guidance on whether you may be a candidate for ReActiv8.
"*" indicates required fields
ReActiv8 is the first and only restorative treatment option for mechanical chronic low back pain as a result of multifidus dysfunction, and aims to restore the neuromuscular control between the brain and spine.

9 out of 10 patients were “Definitely Satisfied” with ReActiv8.
8 out of 10 patients reported substantial (>50%) improvements in pain and/or function.
7 out of 10 patients voluntarily eliminated or reduced their opioid intake*
*Patients that were on opioids at baseline: (1-year=31/65), (2-Year= 34/57), (3-Year= 36/51), (5-Year=36/52)
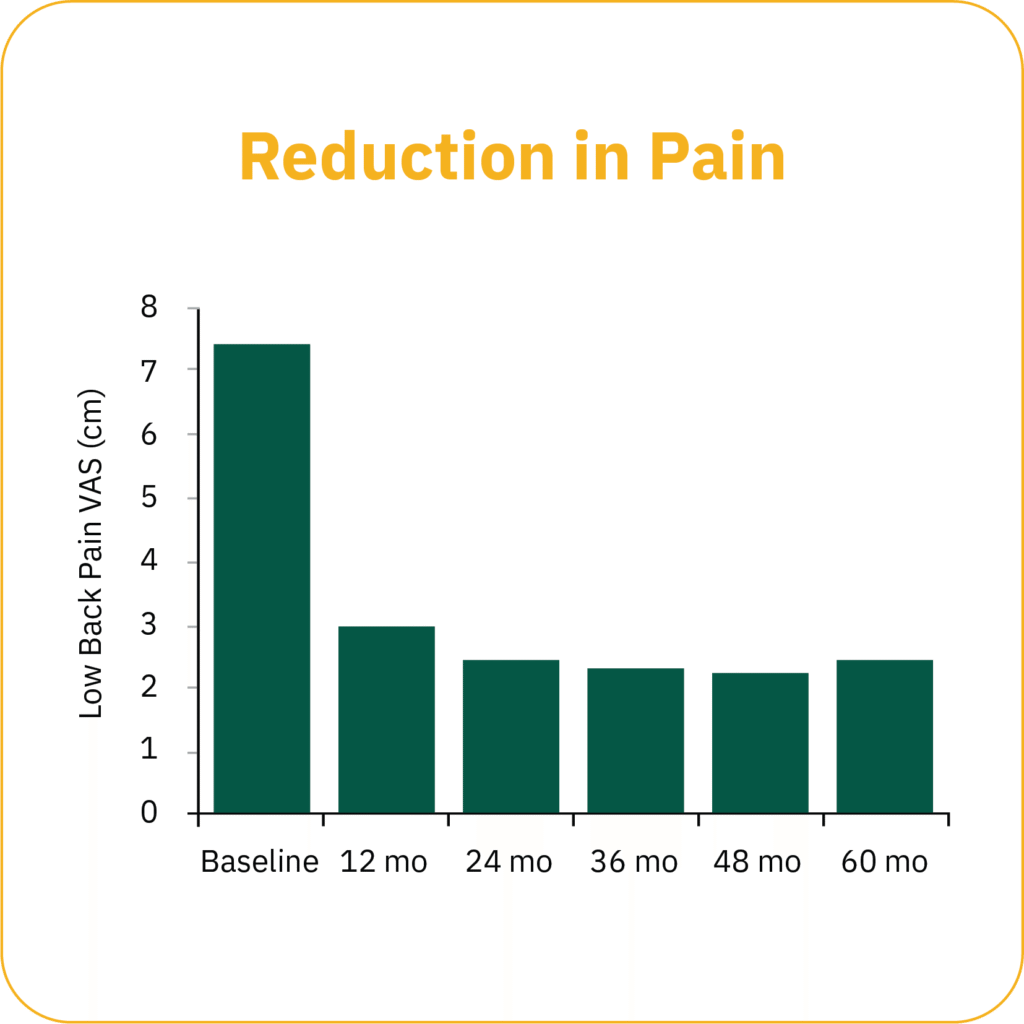
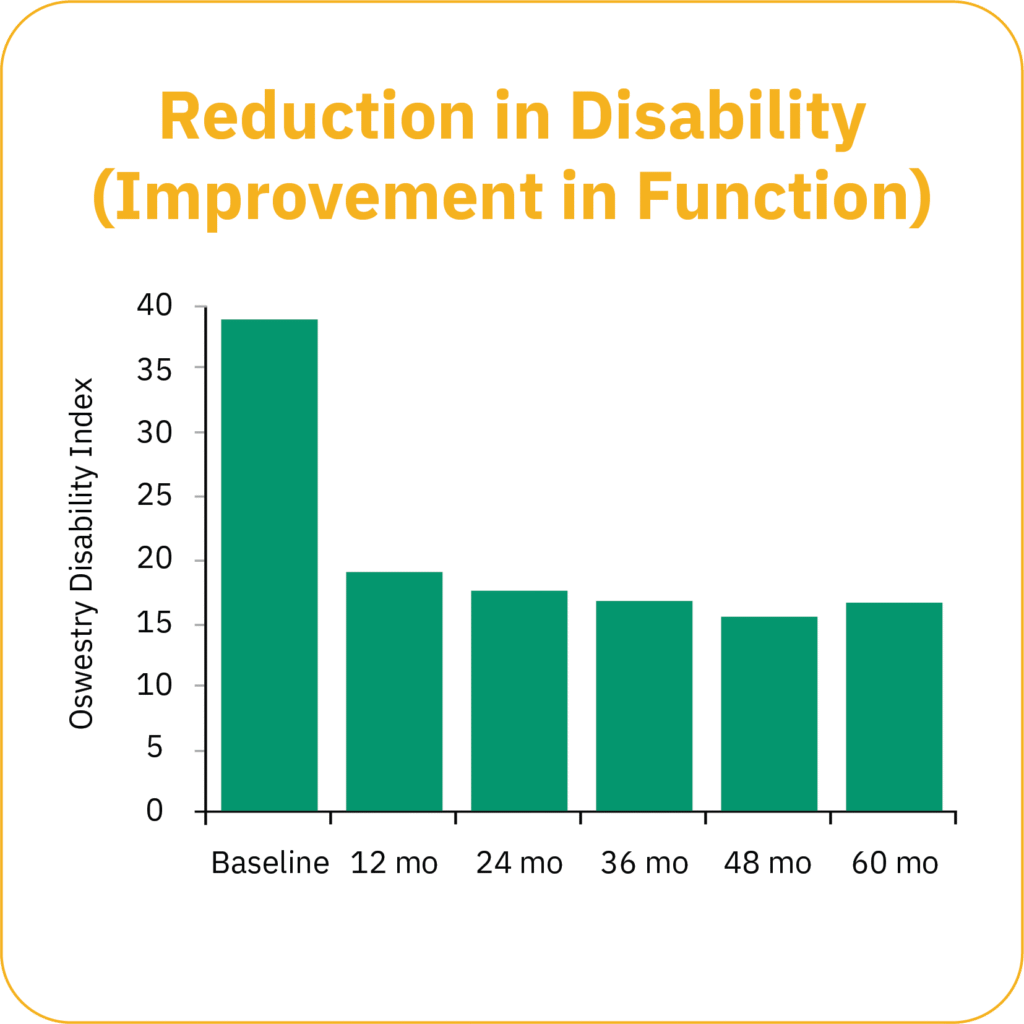
Clinical study data show that patients experience progressive long-term improvements in pain and function, over time, demonstrating a durable and consistent result.*
*Gilligan et al. Five-Year Longitudinal Follow-up of Restorative Neurostimulation Demonstrates Durability of Effectiveness in Patients with Refractory Chronic Low Back Pain associated with Multifidus Muscle Dysfunction
Many people who were treated for their mechanical chronic low back pain with ReActiv8 have seen a dramatic improvement and were eager to share their stories to give hope to others
Learn more about the process here

Hear physician and patient perspectives on mechanical chronic low back pain, the role of the multifidus muscle, and the clinical outcomes on Restorative Neurostimulation Therapy.
Our dedicated patient access specialists will work with you, your physician, and your health insurance company in an effort to obtain authorization for the therapy.
FAQS
ReActiv8 is designed to address the underlying cause of mechanical low back pain by helping patients restore neuromuscular control of the multifidus muscle.
The multifidus muscle is the key stabilizing muscle in the lower back. A single injury to the back can cause impaired muscle control of the multifidus. This lack of muscle control can decrease the spine’s functional stability, leaving the spine susceptible to further injury and overloading. Restoring the neuromuscular control of the multifidus muscle can stabilize the spine, therebyincreasing function and decreasing pain.
By enrolling in the RSVP Program, you may be able to increase your access to ReActiv8 through the submission of prior authorization and patient-based appeals.
Enroll in the RSVP program here.
ReActiv8 addresses mechanical CLBP, delivers stimulation to the multifidus muscle in two 30-min sessions daily, and aims to restore neuromuscular control to increase function. SCS addresses neuropathic CLBP, delivers stimulation 24/7, and aims to interfere with the perception of pain.
Talk to you doctor to see if you are a candidate.
Take the health survey and get scheduled for a consultation to see if you are a candidate.